Ammonium sulphate is a widely used chemical compound in various industries, including agriculture, pharmaceuticals, and food processing. Understanding its properties, particularly its Ammonium Sulphate Molecular Mass, is crucial for its effective application. This blog post delves into the molecular mass of ammonium sulphate, its chemical properties, applications, and safety considerations.
Understanding Ammonium Sulphate
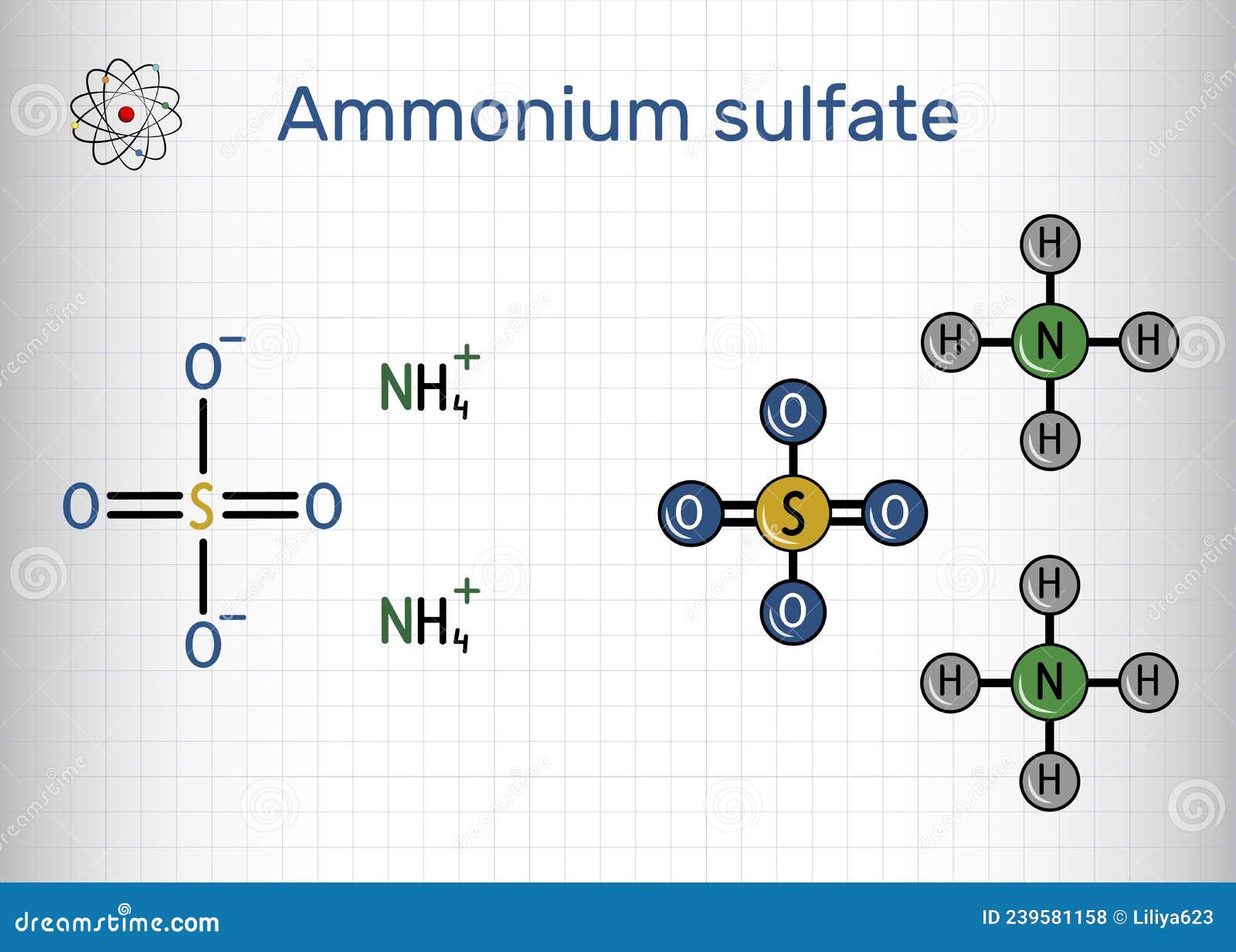
Ammonium sulphate, with the chemical formula (NH4)2SO4, is a white crystalline salt that is highly soluble in water. It is commonly used as a fertilizer due to its high nitrogen content, which is essential for plant growth. The compound is also used in the production of other chemicals, in water treatment, and as a food additive.
Calculating the Ammonium Sulphate Molecular Mass
The Ammonium Sulphate Molecular Mass can be calculated by summing the atomic masses of all the atoms in the formula. The atomic masses of the elements involved are as follows:
- Nitrogen (N): 14.01 g/mol
- Hydrogen (H): 1.01 g/mol
- Sulphur (S): 32.07 g/mol
- Oxygen (O): 16.00 g/mol
The molecular formula (NH4)2SO4 indicates that there are:
- 2 nitrogen atoms
- 8 hydrogen atoms
- 1 sulphur atom
- 4 oxygen atoms
Using these values, the Ammonium Sulphate Molecular Mass is calculated as follows:
| Element | Number of Atoms | Atomic Mass (g/mol) | Total Mass (g/mol) |
|---|---|---|---|
| Nitrogen (N) | 2 | 14.01 | 28.02 |
| Hydrogen (H) | 8 | 1.01 | 8.08 |
| Sulphur (S) | 1 | 32.07 | 32.07 |
| Oxygen (O) | 4 | 16.00 | 64.00 |
| Total Molecular Mass | 132.17 | ||
Therefore, the Ammonium Sulphate Molecular Mass is approximately 132.17 g/mol.
📝 Note: The molecular mass is a theoretical value and may vary slightly due to isotopic variations and measurement errors.
Chemical Properties of Ammonium Sulphate
Ammonium sulphate exhibits several key chemical properties that make it valuable in various applications:
- Solubility: It is highly soluble in water, making it easy to dissolve and apply in liquid form.
- pH Level: It is slightly acidic, with a pH of around 5.5 in solution, which can help lower the pH of alkaline soils.
- Stability: It is stable under normal conditions but decomposes upon heating, releasing ammonia and sulphuric acid.
- Hydroscopic Nature: It absorbs moisture from the air, which can affect its storage and handling.
Applications of Ammonium Sulphate
Ammonium sulphate has a wide range of applications across different industries:
Agriculture
In agriculture, ammonium sulphate is primarily used as a fertilizer. It provides both nitrogen and sulphur, which are essential nutrients for plant growth. The nitrogen in ammonium sulphate is in the ammonium form, which is readily available to plants. The sulphur content helps in the production of proteins and enzymes, promoting overall plant health.
Pharmaceuticals
In the pharmaceutical industry, ammonium sulphate is used in the purification of proteins and antibodies. It acts as a precipitating agent, helping to isolate and purify biological molecules. This application is crucial in the production of vaccines, enzymes, and other biopharmaceuticals.
Food Processing
Ammonium sulphate is used as a food additive, particularly in bread making. It acts as a dough conditioner, improving the texture and volume of the bread. It is also used in the production of certain cheeses and as a yeast nutrient.
Water Treatment
In water treatment, ammonium sulphate is used to control the pH of water and to remove impurities. It helps in the precipitation of metals and other contaminants, making the water safer for consumption and industrial use.
Industrial Applications
Ammonium sulphate is used in various industrial processes, including the production of other chemicals, such as ammonium persulphate and ammonium nitrate. It is also used in the manufacture of textiles, leather, and paper.
Safety Considerations
While ammonium sulphate is generally safe to handle, certain precautions should be taken to ensure safety:
- Handling: Wear appropriate personal protective equipment, including gloves and safety glasses, when handling ammonium sulphate.
- Storage: Store in a cool, dry place away from incompatible substances, such as strong oxidizing agents.
- Disposal: Dispose of ammonium sulphate according to local regulations to prevent environmental contamination.
- Health Hazards: Prolonged exposure to ammonium sulphate can cause skin and eye irritation. Inhalation of dust can lead to respiratory issues.
📝 Note: Always refer to the Material Safety Data Sheet (MSDS) for specific safety guidelines and emergency procedures.
Ammonium sulphate is a versatile compound with numerous applications in various industries. Understanding its Ammonium Sulphate Molecular Mass and chemical properties is essential for its effective use. Whether in agriculture, pharmaceuticals, or industrial processes, ammonium sulphate plays a crucial role in enhancing productivity and efficiency. By following proper handling and safety guidelines, its benefits can be maximized while minimizing potential risks.
Related Terms:
- ammonium sulfate nh4 2so4
- boiling point of ammonium sulfate
- molecular weight of ammonium sulphate
- ammonium sulfate common name
- molar mass of ammonium sulfide
- molecular weight ammonium sulfate